When most people hear the word lectin, they think about food lists. Beans. Grains. Nightshades. Seeds. Maybe they think about pressure cookers, soaking methods, or the debate over whether lectins are misunderstood or dangerous. But behind all of those conversations is something much smaller and much more specific. It is the reason lectins matter in the first place. That reason is binding.
Lectins are not just present in foods. They are proteins with a very particular skill. They bind to carbohydrates. Not sugars in the abstract sense, but specific sugar structures that sit on the surface of cells throughout the body. To understand lectins, you have to understand what they are binding to, why that binding happens, and what it can trigger inside the body. Let’s walk through this slowly and simply.
What Is a Binding Site?
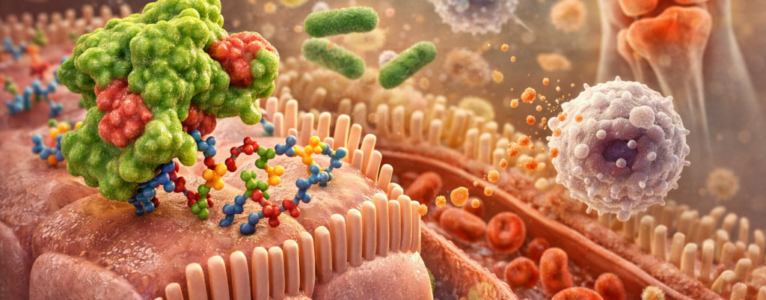
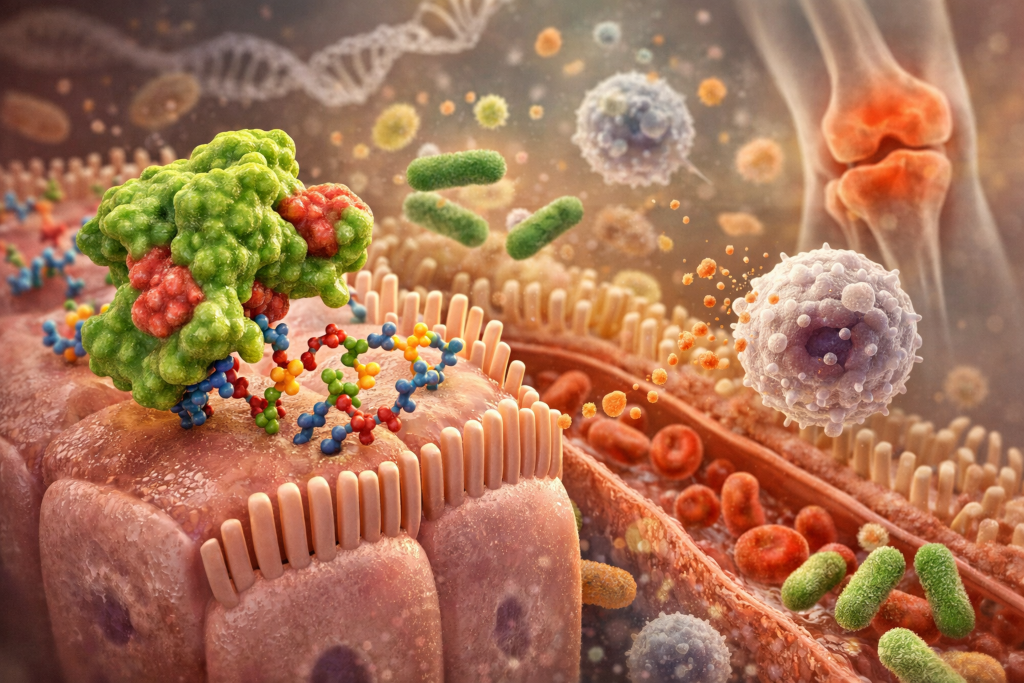
Imagine every cell in your body wearing a coat. That coat is not made of fabric. It is made of molecules. Many of those molecules are glycoproteins and glycolipids, which means they have carbohydrate chains attached to them. These sugar chains stick out from the cell surface like tiny branches. Those branches are not decorative. They are information.
Cells use these carbohydrate structures to communicate. The immune system reads them. Hormones interact with them. Microbes recognize them. These carbohydrate patterns help determine whether something is “self,” “foreign,” inflamed, calm, stressed, or healing.
A binding site is simply a region on a molecule where something else can attach. In the case of lectins, the lectin protein has a structure that allows it to attach to very specific carbohydrate patterns. It is a little like a key that fits certain locks. Not all locks. Only certain ones.
Different lectins recognize different sugar structures. Some bind to mannose. Others bind to N-acetylglucosamine. Others recognize galactose. The specificity matters. It determines where in the body that lectin is most likely to attach and what effects it may have.
Why Plants Make Lectins
Plants produce lectins as part of their defense systems. They cannot run away from insects, fungi, or animals. So they rely on chemical tools. Lectins can bind to carbohydrate structures in the digestive tracts of insects and animals. In some cases, this binding interferes with nutrient absorption or damages gut lining cells. That discourages predators from continuing to eat the plant. From a plant’s perspective, lectins are protective proteins.
From a human perspective, most lectins are harmless when properly prepared. Many are deactivated by soaking, sprouting, fermenting, or pressure cooking. But some lectins are more resistant to digestion. When they survive cooking and digestive enzymes, they can reach the lining of the gut intact. That is where binding becomes relevant to human health.
The Gut Lining: A Busy Surface
Your intestinal lining is not a smooth tube. It is a highly folded, highly active interface between the outside world and your internal systems. It is covered in microvilli, mucus, immune cells, and carbohydrate-rich surface proteins. These carbohydrate structures are normal and necessary. They help regulate what passes through the gut barrier. They support beneficial bacteria. They coordinate immune responses.
If a lectin survives digestion and reaches the small intestine, it can bind to these carbohydrate structures. When binding happens, several things may occur. First, binding can alter cell signaling. Cells constantly send and receive chemical messages. When a lectin attaches to a carbohydrate receptor, it can change how that receptor behaves. That may influence inflammation pathways, tight junction proteins, or immune activation.
Second, binding may affect intestinal permeability. Tight junctions are the structures that hold intestinal cells together. Certain lectins have been shown in laboratory settings to influence these junctions, potentially increasing permeability under specific conditions. Increased permeability allows larger molecules to cross the gut barrier more easily. For people who are sensitive, this can contribute to immune activation.
It is important to emphasize that effects vary widely between individuals. Genetics, microbiome composition, cooking methods, and overall dietary patterns all influence outcomes.
Lectins and the Immune System
The immune system is built to detect patterns. It recognizes carbohydrate structures on bacteria, viruses, and damaged cells. In fact, the body produces its own lectins, called endogenous lectins, such as mannose-binding lectin. These are part of normal immune defense. So lectins are not inherently foreign or toxic. They are part of biology.
The issue arises when dietary lectins bind in ways that stimulate immune pathways excessively or chronically. If a lectin attaches to a receptor on immune cells in the gut, it may trigger cytokine release. Cytokines are signaling molecules that coordinate inflammation. In a healthy, resilient system, occasional stimulation is manageable. The immune system responds and then settles down. But in people with existing gut barrier dysfunction, autoimmune conditions, or chronic inflammation, additional stimulation may feel more significant.
Modern research is increasingly focused on this interaction. Scientists are not just asking whether lectins are good or bad. They are asking how lectin binding interacts with individual immune landscapes.
The Microbiome Connection
Your gut bacteria also have carbohydrate structures on their surfaces. Lectins can bind to them as well. This can influence microbial populations in subtle ways. Some lectins may inhibit certain bacteria while allowing others to thrive. The microbiome itself can also break down or modify lectins, reducing their binding capacity.
This creates a dynamic relationship. A person with a diverse and stable microbiome may process dietary lectins differently than someone with dysbiosis. Emerging research in microbiome mapping suggests that individual responses to lectins may be partly explained by microbial composition. Two people can eat the same food and have very different physiological outcomes.
Cooking and Binding Strength
Binding strength depends on structure. Lectins have specific three-dimensional shapes that allow them to attach to carbohydrate targets. Heat can alter that structure. Traditional food preparation methods often reduce lectin activity significantly. Soaking beans and discarding the soaking water removes some lectins. Pressure cooking denatures many lectin proteins. Fermentation can degrade them enzymatically.
Raw kidney beans contain a lectin called phytohemagglutinin that can cause severe digestive distress if consumed uncooked. Proper boiling destroys it. This is a clear example of how preparation affects binding potential. Not all lectins are equally heat sensitive. Some are more resistant. That is why preparation methods matter more than food categories alone.
Lectin Binding Beyond the Gut
While most discussions focus on digestion, lectin binding can theoretically influence other systems if lectins enter circulation. Under normal conditions, large proteins do not easily pass into the bloodstream. The gut barrier limits this. However, in cases of increased intestinal permeability, small amounts of intact lectins may cross into circulation. If that occurs, they may bind to carbohydrate structures on other cell types.
Research has explored potential interactions with joint tissue, blood vessels, and endocrine signaling. These areas are still under investigation and should be approached with caution. The evidence is evolving, and exaggerated claims often go beyond what the data supports. The key idea is not that lectins travel freely throughout the body in everyone. It is that binding is possible under certain biological conditions.
Individual Sensitivity and Context
One of the most misunderstood aspects of lectins is variability. Some populations have consumed legumes and whole grains for centuries without widespread autoimmune disease. Others report noticeable symptom improvement when reducing high-lectin foods. Both observations can be true.
Binding effects are influenced by:
- Genetic differences in immune receptors
- Microbiome composition
- Integrity of the gut barrier
- Overall inflammatory load
- Cooking and processing methods
When people improve sleep, reduce stress, diversify fiber intake, and support gut health, their tolerance to various foods often changes. Lectins do not operate in isolation. They are part of a larger physiological ecosystem.
What Binding Really Means for Everyday Eating
The concept of binding can sound dramatic, but it is simply molecular interaction. Proteins bind to receptors constantly in the body. Hormones bind. Antibodies bind. Enzymes bind. The question is not whether binding happens. It is what that binding triggers.
For many people, properly prepared legumes, pressure cooked beans, peeled and deseeded nightshades, and fermented grains are tolerated well. For others, reducing certain high-lectin foods during periods of gut repair may provide relief.
Understanding binding sites gives you a framework. It shifts the conversation away from fear and toward mechanism. Instead of thinking, “This food is bad,” you can think, “This protein binds to this structure. Under what conditions might that matter for me?” That is a more empowering question.
Modern Research and Ongoing Questions
Scientists today are using more advanced tools than ever before. Molecular imaging, glycan mapping, and microbiome sequencing allow researchers to observe interactions at finer scales.
Some current areas of interest include:
- How lectin binding influences tight junction proteins
- Whether certain glycan patterns make some individuals more reactive
- How microbiome diversity modifies lectin activity
- The potential role of lectins in metabolic signaling
Importantly, the scientific community does not universally classify dietary lectins as toxins. Many lectin-containing foods also contain fiber, polyphenols, and resistant starch that support metabolic health. The research landscape is nuanced. It recognizes both potential risks and potential benefits depending on context.
Bringing It Back to Practical Living
If you are exploring a low-lectin lifestyle, understanding binding sites helps you make informed decisions without becoming overly restrictive.
You can focus on preparation methods rather than elimination alone. You can pay attention to how your body responds instead of relying solely on generalized rules. You can support gut integrity through sleep, stress management, and balanced nutrition so that occasional lectin exposure is less likely to create problems.
Binding is not a moral issue. It is a biological one. When you zoom in far enough, all of nutrition becomes molecular conversation. Proteins interact with receptors. Carbohydrates signal. Fats influence membranes. Nothing operates in isolation. Lectins are one part of that conversation.
Understanding their binding sites simply means understanding where they attach and what may follow. Once you see that clearly, the topic becomes less mysterious and less extreme. It becomes a matter of context, preparation, and individual biology. And that is where real empowerment begins.